Displaying 61 - 70 of 120

Contrast Measurement Method for Optical See-Through Augmented Reality Head-Mounted Displays
Categories:
Virtual
This regulatory science tool (RST) describes a method to characterize digital image quality on optical see-through augmented reality head-mounted displays (AR HMDs) in ambient lighting conditions. The method can be implemented to measure AR display performance, such as Michelson contrast and contrast uniformity.
Program Areas:
Medical Extended Reality

Tissue and Blood Mimicking Materials for High Intensity Therapeutic Ultrasound
Categories:
Lab Method
This regulatory science tool is a lab method that may be helpful while evaluating high intensity therapeutic ultrasound (HITU) devices.
Program Areas:
Therapeutic Ultrasound

Highly Adaptive Risk Assessment Model (HARAM) App for Retrospective Analysis of COVID-19 Intervention Strategies
Categories:
Computer Model
This regulatory science tool comprises a model for conducting a retrospective analysis of COVID-19 intervention strategies.
Program Areas:
Emergency Preparedness
Method and Phantom Design for the Evaluation of Material Quantification Accuracy of Contrast-Enhanced Spectral Computed Tomography (CT) Systems
This regulatory science tool is a method and phantom design for the evaluation of iodine-specific quantitative measurements, specifically the accurate estimation of iodine concentration within contrast-enhanced spectral CT.
Program Areas:
Medical Imaging and Diagnostics
M-SYNTH: A Dataset for the Comparative Evaluation of Mammography AI
Categories:
Dataset
This regulatory science tool is a synthetic mammography dataset that includes a variety of breast densities, breast sizes, and inserted lesions imaged with different exposure levels. The dataset is intended to be used for the comparative evaluation of AI tools used in mammography.
Program Areas:
AI / Machine Learning

Multifunctional Method for Quantitative Evaluation of Time-Dependent Eye Hazard from Multiwavelength Laser Pointers
Categories:
Lab Method
This regulatory science tool (RST) is a lab method for evaluating the safety of critical time-dependent radiant power, spectral, and temporal characteristics for multiwavelength high-power laser pointers and handheld compact laser products
Program Areas:
Ophthalmology
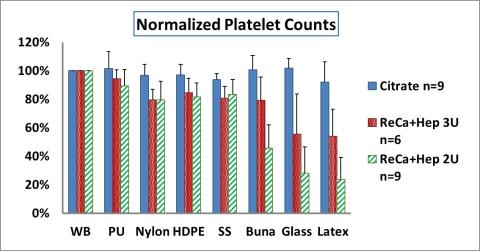
Test considerations for performing platelet and leukocyte count assays for in vitro hemocompatibility assessment of Blood-contacting materials per ASTM F2888-19 standard
Categories:
Lab Method
This regulatory science tool is a lab method for performing platelet and leukocyte count assays per the <a href="https://www.iso.org/standard/63448.html">ASTM F2888-19 standard</a>, as a part of in vitro thrombogenicity assessment of blood-contacting medical device materials.
Program Areas:
Cardiovascular

Head Mounted Display Eye-box Centering using Transverse Chromatic Aberrations
Categories:
Lab Method
This regulatory science tool (RST) is a method for assessing the image quality of a head mounted display using the display’s transverse chromatic aberration characteristics to align the light measuring device to the eye-point/ center of the eye-box.
Program Areas:
Medical Extended Reality
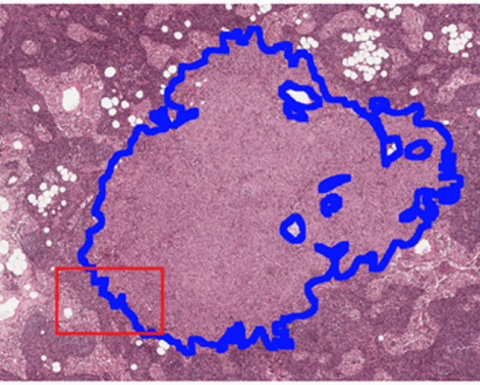
ValidPath: Whole Slide Image Processing and Machine Learning Performance Assessment Tool
Categories:
Computer Model
This regulatory science tool is a software program written in Python for analyzing whole slide images (WSIs) and assisting developers or pathologists in the assessment of machine learning algorithms used in digital pathology.

TRUST: A Testbed Design Model for Evaluating 5G-Enabled Medical Devices
Categories:
Lab Method
This regulatory science tool is a lab method for building a 5G network testbed to assess data transmission of medical device functions using 5G connectivity.
Program Areas:
Electromagnetic and Electrical Safety
