Catalog of Regulatory Science Tools to Help Assess New Medical Devices
This regulatory science tool (RST) is a phantom that enables image quality assessment of ophthalmic optical coherence tomography (OCT) devices.
Technical Description
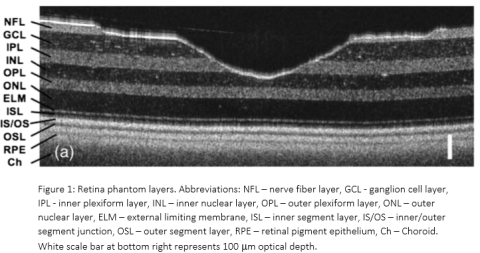
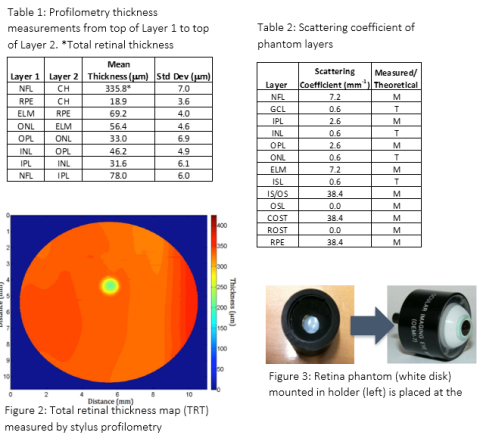
The retina phantom closely approximates the optical scattering and thickness of the many individual layers of the retina. It is made of thin layers of silicone (polydimethylsiloxane) with different amounts and types of micro- or nanoparticles added to each layer. The phantom also includes surface topography to mimic the center of the retina (macula), namely a foveal pit. As shown in the image from a laboratory OCT system in Figure 1, this phantom yields a cross-sectional OCT B-scan image similar to what is acquired from a human retina. The thickness of each retinal layer was measured independently via stylus profilometry (Table 1) and the optical scattering coefficient of each layer was either measured via spectrophotometry or estimated with the documented optical properties of silicone and scattering particles (Table 2). A map of the overall thickness of the phantom is shown in Figure 2. The phantom is enclosed in an off-the-shelf model eye (Ocular Instruments OEMI-7, Figure 3), replacing the existing model retina at the back of the eye.
Intended Purpose
This retina phantom with model eye is intended to be a convenient bench testing tool for assessing imaging performance of OCT devices designed to acquire images of the human retina in vivo.
Testing
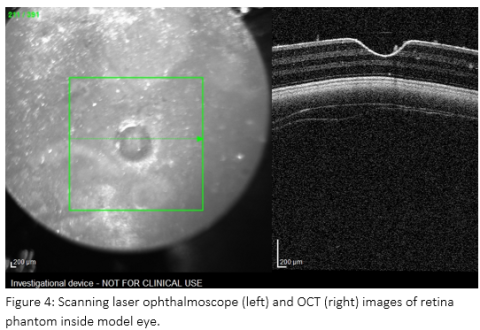
The retina phantom was tested qualitatively via imaging the model eye with an investigational version of a clinical ophthalmic OCT device, Heidelberg Spectralis. This device includes a scanning laser ophthalmoscope which produces a surface image of the retina covering the region imaged with OCT. A representative image of the phantom from this device is shown in Figure 4.
Limitations
Because the retina phantom does not fully recreate the detailed structure of both healthy and diseased human retinas, an OCT layer segmentation algorithm cannot be fully tested and clinical data remains necessary to establish performance for regulatory purposes.
Supporting Documentation
Peer-reviewed journal article describing the retina phantom: https://doi.org/10.1117/1.JBO.19.2.021106
Contact
Tool Reference
- RST Reference Number: RST26OM01.01
- Date of Publication: 05/05/2026
- Recommended Citation: U.S. Food and Drug Administration. (2026). Retina Phantom with Model Eye for Ophthalmic Optical Coherence Tomography (OCT) Devices (RST26OM01.01). https://cdrh-rst.fda.gov/retina-phantom-model-eye-ophthalmic-optical-coherence-tomography-oct-devices
